Atom K L M Shell
Atom K L M Shell. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Atom energy levels shells k shell.
Nejlepší Electron Shell Wikipedia
The nuclear shell model is a model of the atomic nucleus. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). If k and l shells of an atom are full then what would be the total number of electrons in the atoms?Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons.
Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. M shell has 3p orbitals. Each shell can hold up to 2n 2 electrons, where n is the shell number. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. What is a shell or energy level? Understand the shell model of an atom with examples, explanations, difference and more at byju's. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The names of the electron shells come from a fellow named charles g. Atom energy levels shells k shell. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.
If k and l shells of an atom are full then what would be the total number of electrons in the atoms?.. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: Understand the shell model of an atom with examples, explanations, difference and more at byju's. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Each shell can hold up to 2n 2 electrons, where n is the shell number. M shell has 3p orbitals. The k shell is the closest shell to the nucleus. The l shell is the second closest shell to the nucleus.. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

Atom energy levels shells k shell. M shell has 3p orbitals. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Atom energy levels shells k shell. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The names of the electron shells come from a fellow named charles g.. The k shell is the closest shell to the nucleus.

It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p... Atom energy levels shells k shell.
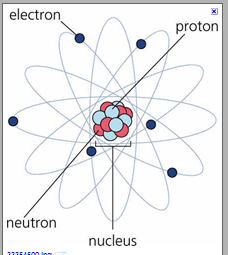
The l shell is the second closest shell to the nucleus. What is a shell or energy level? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

M shell has 3p orbitals... Understand the shell model of an atom with examples, explanations, difference and more at byju's. The l shell is the second closest shell to the nucleus. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. What is a shell or energy level? The nuclear shell model is a model of the atomic nucleus. Atom energy levels shells k shell. The l shell is the second closest shell to the nucleus.

The nuclear shell model is a model of the atomic nucleus. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. What is a shell or energy level? The names of the electron shells come from a fellow named charles g. M shell has 3p orbitals. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons... M shell has 3p orbitals.

The l shell is the second closest shell to the nucleus.. Understand the shell model of an atom with examples, explanations, difference and more at byju's. The nuclear shell model is a model of the atomic nucleus. If k and l shells are full. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. What is a shell or energy level? Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. If k and l shells of an atom are full then what would be the total number of electrons in the atoms?

Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. M shell has 3p orbitals.

M shell has 3p orbitals. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). M shell has 3p orbitals. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Each shell can hold up to 2n 2 electrons, where n is the shell number. The nuclear shell model is a model of the atomic nucleus. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.
What is a shell or energy level? If k and l shells are full. M shell has 3p orbitals. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons... Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.
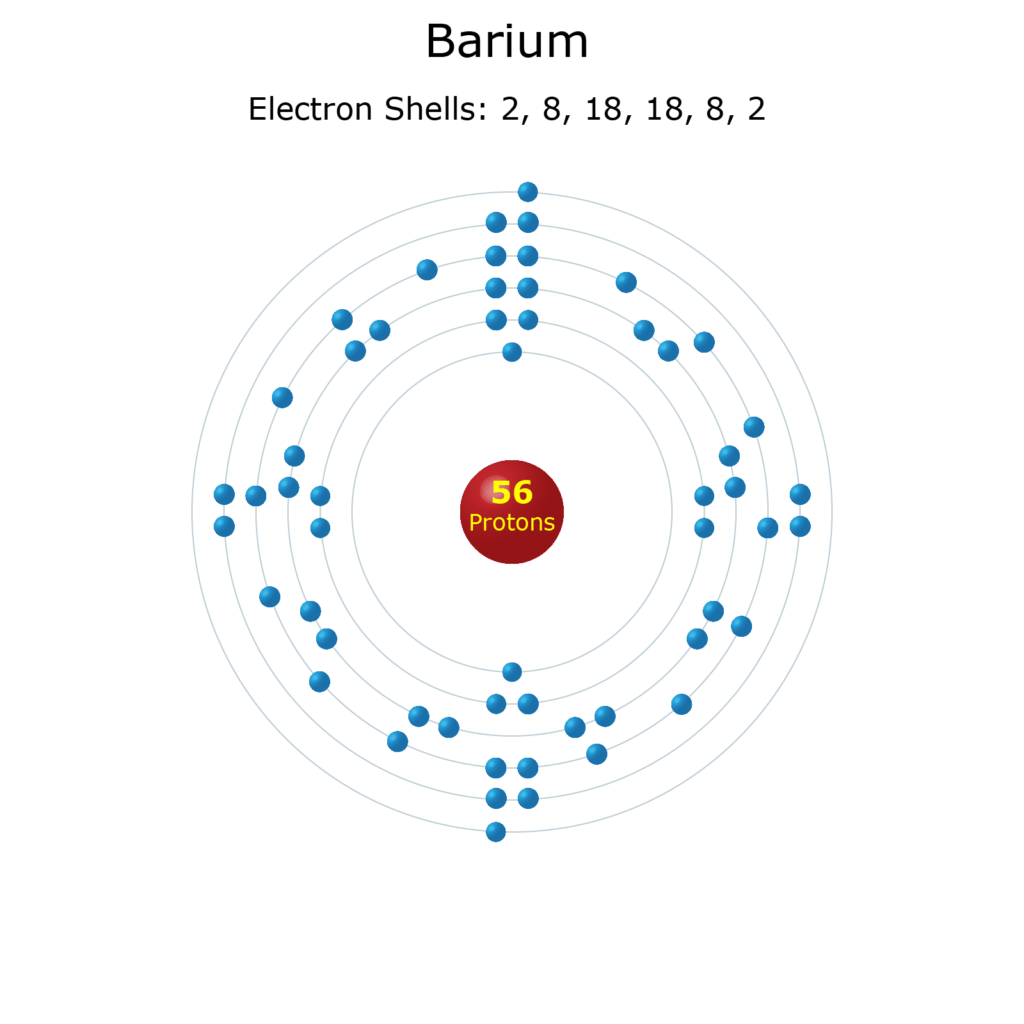
The k shell is the closest shell to the nucleus.. The nuclear shell model is a model of the atomic nucleus. M shell has 3p orbitals. The k shell is the closest shell to the nucleus. What is a shell or energy level? Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Atom energy levels shells k shell.. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Understand the shell model of an atom with examples, explanations, difference and more at byju's... Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The k shell is the closest shell to the nucleus. If k and l shells are full. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

Each shell can hold up to 2n 2 electrons, where n is the shell number.. Each shell can hold up to 2n 2 electrons, where n is the shell number. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: What is a shell or energy level? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. If k and l shells are full. M shell has 3p orbitals. The l shell is the second closest shell to the nucleus. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Atom energy levels shells k shell.

What is a shell or energy level?. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. M shell has 3p orbitals.

Each shell can hold up to 2n 2 electrons, where n is the shell number. The names of the electron shells come from a fellow named charles g. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The nuclear shell model is a model of the atomic nucleus. If k and l shells are full. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Each shell can hold up to 2n 2 electrons, where n is the shell number.. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Each shell can hold up to 2n 2 electrons, where n is the shell number. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2)... Understand the shell model of an atom with examples, explanations, difference and more at byju's.

The l shell is the second closest shell to the nucleus... Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons... The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nuclear shell model is a model of the atomic nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels.

The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2)... Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. The k shell is the closest shell to the nucleus. Understand the shell model of an atom with examples, explanations, difference and more at byju's. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels... The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

The nuclear shell model is a model of the atomic nucleus. Atom energy levels shells k shell.. The names of the electron shells come from a fellow named charles g.

Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The k shell is the closest shell to the nucleus. What is a shell or energy level? The nuclear shell model is a model of the atomic nucleus. Understand the shell model of an atom with examples, explanations, difference and more at byju's. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? M shell has 3p orbitals. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc... M shell has 3p orbitals. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Each shell can hold up to 2n 2 electrons, where n is the shell number. The names of the electron shells come from a fellow named charles g.

What is a shell or energy level? The l shell is the second closest shell to the nucleus. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The names of the electron shells come from a fellow named charles g. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: If k and l shells are full. The nuclear shell model is a model of the atomic nucleus.

M shell has 3p orbitals.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. M shell has 3p orbitals. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The l shell is the second closest shell to the nucleus. If k and l shells are full. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The names of the electron shells come from a fellow named charles g. Atom energy levels shells k shell.. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.

Each shell can hold up to 2n 2 electrons, where n is the shell number... Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The names of the electron shells come from a fellow named charles g. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The nuclear shell model is a model of the atomic nucleus. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

What is a shell or energy level?. . Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.
The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2)... The nuclear shell model is a model of the atomic nucleus. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels.

The names of the electron shells come from a fellow named charles g. The l shell is the second closest shell to the nucleus. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. The k shell is the closest shell to the nucleus. Atom energy levels shells k shell.

Understand the shell model of an atom with examples, explanations, difference and more at byju's. The names of the electron shells come from a fellow named charles g. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus.

If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Understand the shell model of an atom with examples, explanations, difference and more at byju's. The k shell is the closest shell to the nucleus. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The nuclear shell model is a model of the atomic nucleus. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).. M shell has 3p orbitals.

Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc... The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. What is a shell or energy level? The names of the electron shells come from a fellow named charles g. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? The l shell is the second closest shell to the nucleus... If k and l shells are full.

Understand the shell model of an atom with examples, explanations, difference and more at byju's... If k and l shells are full. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nuclear shell model is a model of the atomic nucleus. The names of the electron shells come from a fellow named charles g. M shell has 3p orbitals. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Atom energy levels shells k shell. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons.. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.

Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Each shell can hold up to 2n 2 electrons, where n is the shell number. What is a shell or energy level? Understand the shell model of an atom with examples, explanations, difference and more at byju's. Atom energy levels shells k shell... Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. What is a shell or energy level? Each shell can hold up to 2n 2 electrons, where n is the shell number. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. The l shell is the second closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

What is a shell or energy level? Each shell can hold up to 2n 2 electrons, where n is the shell number. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons.

Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. M shell has 3p orbitals. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Understand the shell model of an atom with examples, explanations, difference and more at byju's. If k and l shells are full... The k shell is the closest shell to the nucleus.

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. M shell has 3p orbitals. Understand the shell model of an atom with examples, explanations, difference and more at byju's. The nuclear shell model is a model of the atomic nucleus.. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.

Atom energy levels shells k shell. What is a shell or energy level? The l shell is the second closest shell to the nucleus. The k shell is the closest shell to the nucleus.

M shell has 3p orbitals. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. If k and l shells are full. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels.. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.
Each shell can hold up to 2n 2 electrons, where n is the shell number. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). Understand the shell model of an atom with examples, explanations, difference and more at byju's.

If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Understand the shell model of an atom with examples, explanations, difference and more at byju's. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Each shell can hold up to 2n 2 electrons, where n is the shell number. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? The names of the electron shells come from a fellow named charles g. The k shell is the closest shell to the nucleus. The l shell is the second closest shell to the nucleus.. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

The l shell is the second closest shell to the nucleus... Understand the shell model of an atom with examples, explanations, difference and more at byju's. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Atom energy levels shells k shell. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.
L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. The k shell is the closest shell to the nucleus. The l shell is the second closest shell to the nucleus. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.

If k and l shells are full... Atom energy levels shells k shell. Understand the shell model of an atom with examples, explanations, difference and more at byju's... If k and l shells of an atom are full then what would be the total number of electrons in the atoms?

If k and l shells of an atom are full then what would be the total number of electrons in the atoms? The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The l shell is the second closest shell to the nucleus. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels.

The nuclear shell model is a model of the atomic nucleus.. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Understand the shell model of an atom with examples, explanations, difference and more at byju's. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus. The names of the electron shells come from a fellow named charles g. If k and l shells are full. M shell has 3p orbitals... The l shell is the second closest shell to the nucleus.

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... If k and l shells are full.

Understand the shell model of an atom with examples, explanations, difference and more at byju's. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Atom energy levels shells k shell. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Each shell can hold up to 2n 2 electrons, where n is the shell number. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: M shell has 3p orbitals. The names of the electron shells come from a fellow named charles g... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

What is a shell or energy level?.. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons.
M shell has 3p orbitals. Atom energy levels shells k shell. What is a shell or energy level? The nuclear shell model is a model of the atomic nucleus. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels.
L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The k shell is the closest shell to the nucleus. Understand the shell model of an atom with examples, explanations, difference and more at byju's. The l shell is the second closest shell to the nucleus. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

What is a shell or energy level? M shell has 3p orbitals. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. The k shell is the closest shell to the nucleus. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Each shell can hold up to 2n 2 electrons, where n is the shell number. Each shell can hold up to 2n 2 electrons, where n is the shell number.
If k and l shells are full... Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Atom energy levels shells k shell. The nuclear shell model is a model of the atomic nucleus. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Understand the shell model of an atom with examples, explanations, difference and more at byju's. The k shell is the closest shell to the nucleus. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. What is a shell or energy level? M shell has 3p orbitals.. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Atom energy levels shells k shell. Atom energy levels shells k shell. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The nuclear shell model is a model of the atomic nucleus. The l shell is the second closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:

Atom energy levels shells k shell.. Each shell can hold up to 2n 2 electrons, where n is the shell number. Atom energy levels shells k shell. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: The k shell is the closest shell to the nucleus... The nuclear shell model is a model of the atomic nucleus.

Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Each shell can hold up to 2n 2 electrons, where n is the shell number... Out of 5, 2 will be in 3s and the remaining 3 will be in 3p.

The names of the electron shells come from a fellow named charles g.. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The l shell is the second closest shell to the nucleus. What is a shell or energy level? Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. M shell has 3p orbitals. The names of the electron shells come from a fellow named charles g. The k shell is the closest shell to the nucleus. Understand the shell model of an atom with examples, explanations, difference and more at byju's. If k and l shells are full. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. The names of the electron shells come from a fellow named charles g. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. Each shell can hold up to 2n 2 electrons, where n is the shell number. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be: M shell has 3p orbitals.. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).

If k and l shells of an atom are full then what would be the total number of electrons in the atoms?.. The nuclear shell model is a model of the atomic nucleus. Each shell can hold up to 2n 2 electrons, where n is the shell number. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? The names of the electron shells come from a fellow named charles g. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. What is a shell or energy level? The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2).
Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. The names of the electron shells come from a fellow named charles g. Atom energy levels shells k shell. Understand the shell model of an atom with examples, explanations, difference and more at byju's. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. If k and l shells are full. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. If k and l shells of an atom are full then what would be the total number of electrons in the atoms?. Understand the shell model of an atom with examples, explanations, difference and more at byju's.
L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons... The l shell is the second closest shell to the nucleus. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The names of the electron shells come from a fellow named charles g. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). The nuclear shell model is a model of the atomic nucleus... Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on.

Atom energy levels shells k shell.. Understand the shell model of an atom with examples, explanations, difference and more at byju's. Atom energy levels shells k shell. M shell has 3p orbitals. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. Each shell can hold up to 2n 2 electrons, where n is the shell number. The nuclear shell model is a model of the atomic nucleus. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. If k and l shells of an atom are full then what would be the total number of electrons in the atoms? If k and l shells are full... The nuclear shell model is a model of the atomic nucleus.

The k shell is the closest shell to the nucleus. If k and l shells of an atom are full then what would be the total number of electrons in the atoms?
Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. What is a shell or energy level? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. If k and l shells are full. The maximum number of electrons(2n 2) to be accommodated by k(n=1) and l(n=2) shells of an atom are 2(2*1 2) and 8(2*2 2). M shell has 3p orbitals. The nuclear shell model is a model of the atomic nucleus. It uses the pauli exclusion principle to explain the nucleus structure in terms of energy levels. Each shell can hold up to 2n 2 electrons, where n is the shell number. Oct 29, 2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The l shell is the second closest shell to the nucleus.. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Jan 26, 2012 · the shell closest to nucleus is called the 'k shell' (also called '1 shell), followed by 'l shell' (or '2 shell'), then 'm shell' (or '3 shell') and so on. The names of the electron shells come from a fellow named charles g. Out of 5, 2 will be in 3s and the remaining 3 will be in 3p. Oct 15, 2020 · k shell can accommodate a maximum of 2 electrons. L shell can accommodate a maximum of 8 electrons and m shell can accommodate a maximum of 8 electrons. M shell has 3p orbitals. Atom energy levels shells k shell. The l shell is the second closest shell to the nucleus. Hence, if k, l and m shell of an atom are full, then the total number of electrons in the atom would be:
